
C57BL/6-Glp1rtm1(GLP1R)Bcgen/Bcgen • 170164
このページで
GLP1R (Glucagon-like peptide-1 receptor) is a G protein–coupled receptor (GPCR) highly expressed in pancreatic β-cells and critical for glucose-dependent insulin secretion, appetite regulation, and energy balance. GLP1R is the therapeutic target of GLP-1 receptor agonists (GLP-1RAs), such as semaglutide and liraglutide, which are widely used for the treatment of type 2 diabetes and obesity.
In GLP1R humanized mice, the murine Glp1r gene is replaced with the human GLP1R gene. Homozygous mice express human GLP1R in physiologically relevant tissues without altering immune or metabolic homeostasis. This model provides a translationally relevant platform for preclinical efficacy testing, antibody validation, and drug development targeting GLP1R.
GLP1R humanized mice are an ideal preclinical model for evaluating GLP1R-targeted therapeutics in metabolic disease research, including type 2 diabetes and obesity.
Key Advantages
Validation
Applications
Gene targeting strategy for B-hGLP1R mice. The full coding sequence of human GLP1R gene was inserted into mouse Glp1r gene exon1 in B-hGLP1R mice. The human GLP1R protein expression will be driven by endogenous mouse Glp1r promoter, while mouse Glp1r gene transcription and translation will be disrupted.
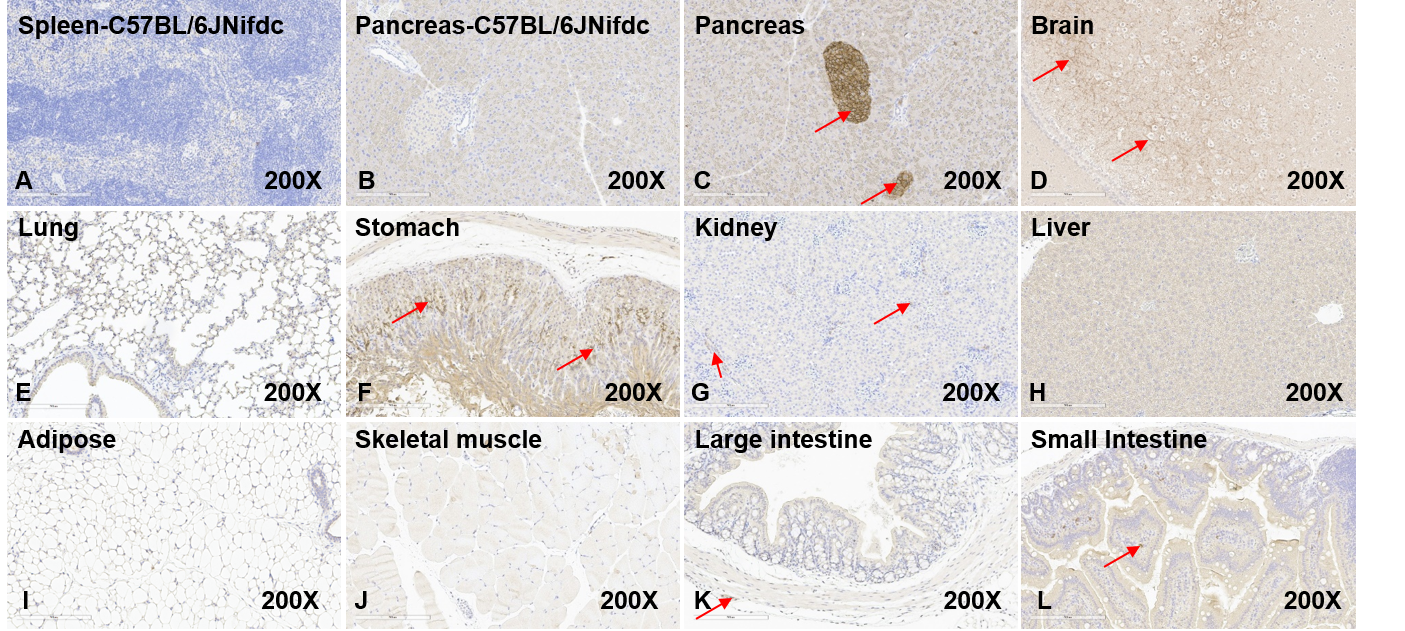
Human GLP1R expression in different tissues of B-hGLP1R mice by IHC. Tissues were stained with human-specific antibodies GLP1R (Anti-GLP1R antibody, abcam, ab254352) (B-L). The spleen of wild-type C57BL/6JNifdc mice was tissue-negative control (A). The Pancreas of C57BL/6JNifdc mice shows human GLP1R negative (B). The islet of the pancreas, striatum septum of brain, gastric mucosal layer and myenteric plexus of stomach, partial blood vessels the kidney, the muscularis propria nerve plexus of both large intestine and small intestine, and the interstitial cells of the small intestine mucosal layer of B-hGLP1R mice show human GLP1R positive (C, D, F, G, K, L). Lung, liver, adipose, and skeletal muscle of B-hGLP1R mice show human GLP1R negative. Original magnification ×200. Abbreviations: IHC, immunohistochemistry.
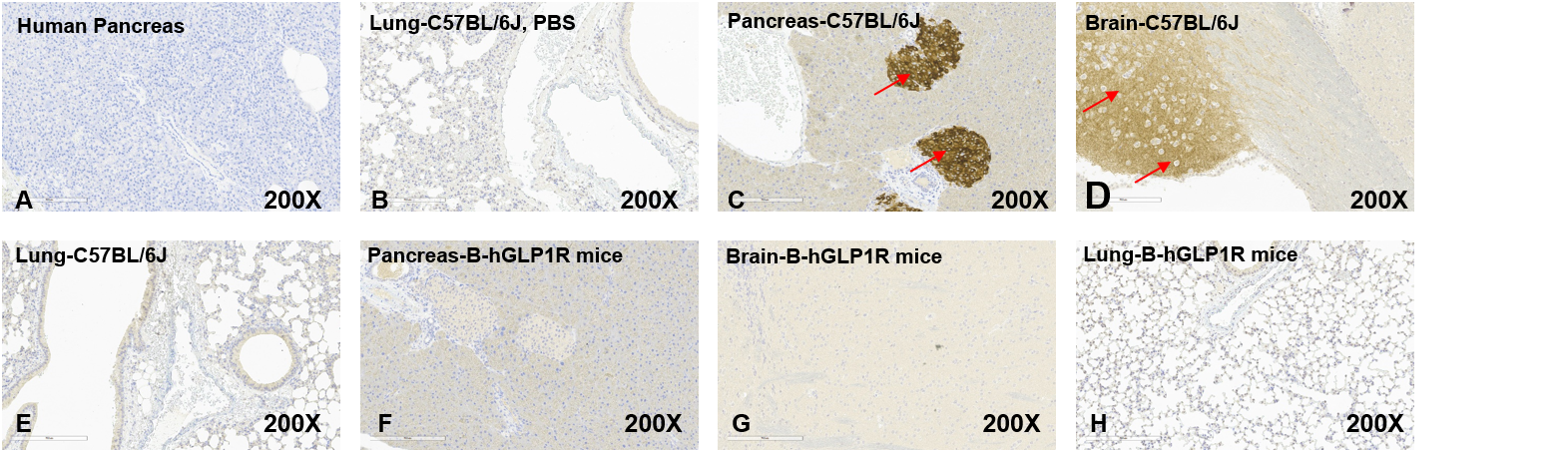
Representative mouse GLP1R expression in different tissues of B-hGLP1R mice by IHC. Tissues were stained with mouse-specific antibodies GLP1R (Anti-GLP-1R antibody, ab218532) (B-H). The human pancreas tissue negative control (A). The pancreas and brain of wild-type C57BL/6JNifdc mice showed mouse GLP1R positive (C, D). The lung tissue of wild-type C57BL/6JNifdc mice showed mouse GLP1R negative (E). The pancreas, brain, and lung of B-hGLP1R mice showed mouse GLP1R negative (F, G, H). Original magnification ×200. Abbreviations: IHC, immunohistochemistry.
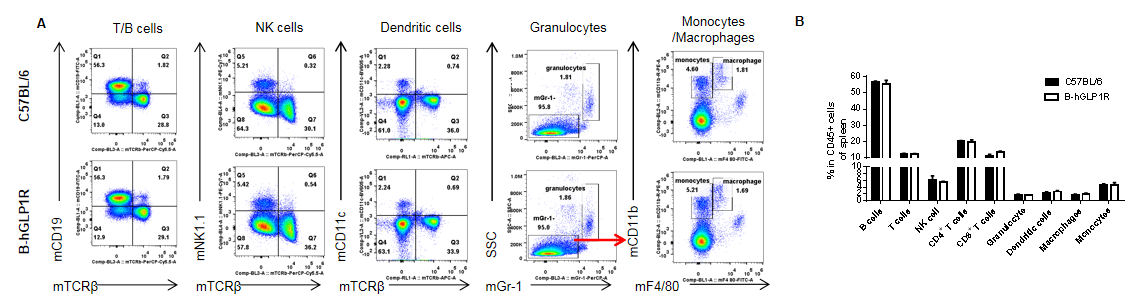
Analysis of spleen leukocyte subpopulations in GLP1R humanized mice. Splenocytes were isolated from female C57BL/6 and GLP1R humanized mice (n=3, 9 weeks old). Flow cytometry was performed to analyze leukocyte subpopulations. Single live cells were gated for the CD45⁺ population and further analyzed by FACS. Representative plots (A) and quantitative results (B) are shown. The percentages of T cells, B cells, NK cells, monocytes, dendritic cells, granulocytes, and macrophages in homozygous GLP1R humanized mice were comparable to those in wild-type C57BL/6 controls. Humanization of GLP1R does not alter the development, differentiation, or distribution of leukocyte subsets in the spleen. Values are expressed as mean ± SEM.
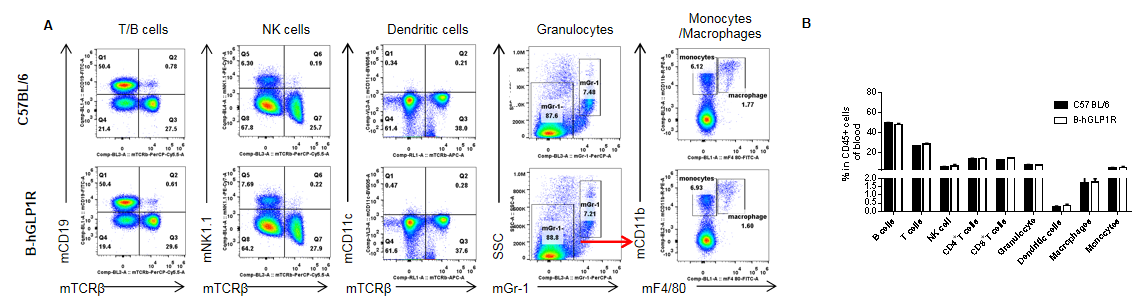
Analysis of blood leukocyte subpopulations in GLP1R humanized mice. Peripheral blood cells were isolated from female C57BL/6 and GLP1R humanized mice (B-hGLP1R, n=3, 9 weeks old). Flow cytometry was performed to evaluate leukocyte subpopulations. Single live cells were gated for the CD45⁺ population and analyzed by FACS. Representative plots (A) and quantification results (B) are shown. The percentages of T cells, B cells, NK cells, monocytes, dendritic cells, granulocytes, and macrophages in homozygous GLP1R humanized mice were comparable to those in C57BL/6 wild-type controls. Humanization of GLP1R does not affect the development, differentiation, or distribution of blood leukocyte subpopulations. Values are expressed as mean ± SEM.
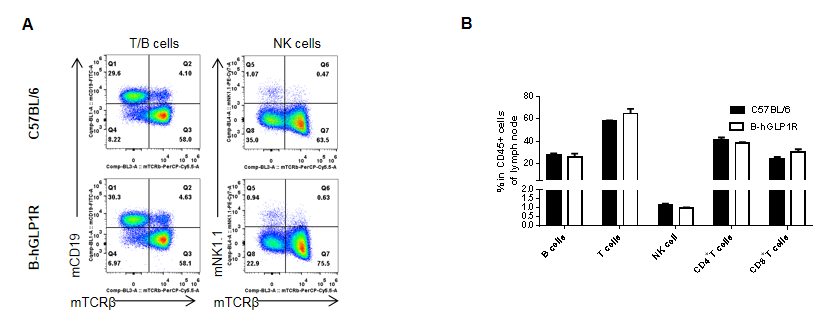
Analysis of lymph node leukocyte subpopulations in GLP1R humanized mice. Lymph nodes were isolated from female C57BL/6 and GLP1R humanized mice (B-hGLP1R, n=3, 9 weeks old). Flow cytometry was performed to evaluate leukocyte subpopulations. Single live cells were gated for the CD45⁺ population and analyzed by FACS. Representative plots (A) and quantitative results (B) are shown. The percentages of T cells, B cells, and NK cells in homozygous GLP1R humanized mice were comparable to those in wild-type C57BL/6 controls. Humanization of GLP1R does not affect the development, differentiation, or distribution of lymph node leukocyte subsets. Values are expressed as mean ± SEM.

Analysis of T cell subpopulations in GLP1R humanized mice. Lymphocytes were isolated from spleen, lymph nodes, and peripheral blood of female C57BL/6 and GLP1R humanized mice (B-hGLP1R, n=3, 9 weeks old). Flow cytometry was performed to assess T cell subpopulations. Cells were gated on the CD45⁺ population and analyzed for CD4⁺, CD8⁺, and regulatory T cell (Treg) subsets. The proportions of CD4⁺ T cells, CD8⁺ T cells, and Tregs in GLP1R humanized mice were comparable to those in wild-type controls across spleen, blood, and lymph nodes. Humanization of GLP1R does not alter the development, differentiation, or distribution of major T cell subsets in immune organs. Values are expressed as mean ± SEM.
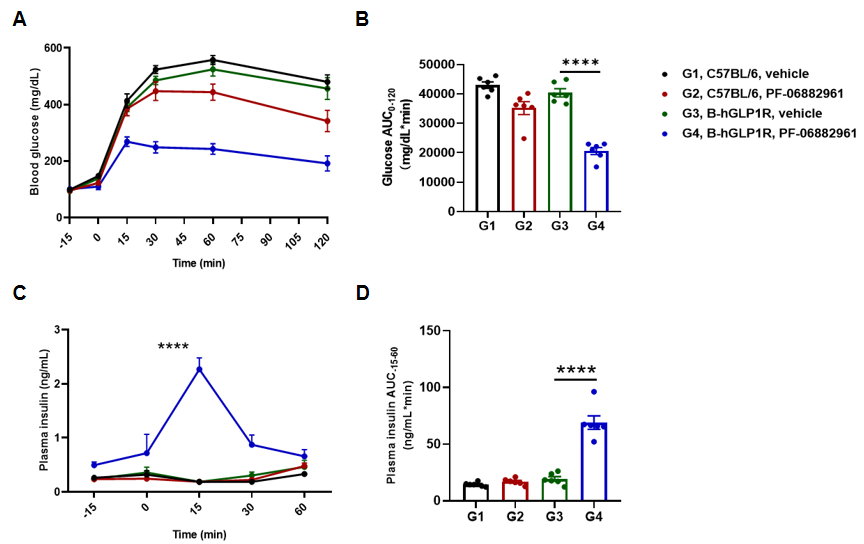
PF-06882961 improved glucose tolerance and promote insulin secretion in B-hGLP1R mice. B-hGLP1R mice were treated with PF-06882961 for 3 mg/kg, s.c. Then the glucose tolerance test was performed in C57BL/6 and B-hGLP1R mice for 2g/kg D-Glucose is administered by intraperitoneal injection (IPGTT). (A) Blood glucose. (B) Area under the curve (AUC) for IPGTT. (C) Plasma insulin. (D) Area under the curve (AUC) for the plasma insulin. As shown, glucose tolerance was improved by PF-06882961 in B-hGLP1R mice but not in wild-type C57BL/6 mice, and promote plasma insulin secretion in B-hGLP-1R mice. Values are expressed as mean ± SEM. Significance was determined by the Unpaired t test. (*P < 0.05, **P < 0.01, ***P < 0.0001)
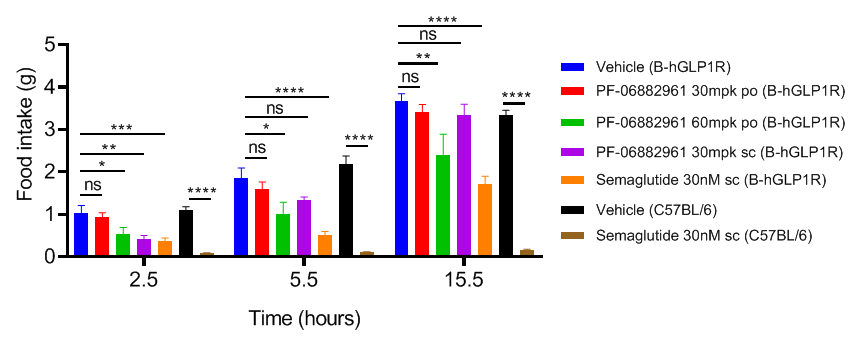
Study of PF-06882961 and Semaglutide in B-hGLP1R mice food intake. B-hGLP1R mice (12-week-old) were acclimated to handing and oral gavage vehicle for 3 days before study. Mice were grouped by body weight and daily food intake. Subsequently, the mice were treated with dose and route shown in the figure. Food weights were measured on 2.5, 5.5, 15.5 hours after treatment. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA . (*P < 0.05, **P < 0.01, ***P < 0.0001)
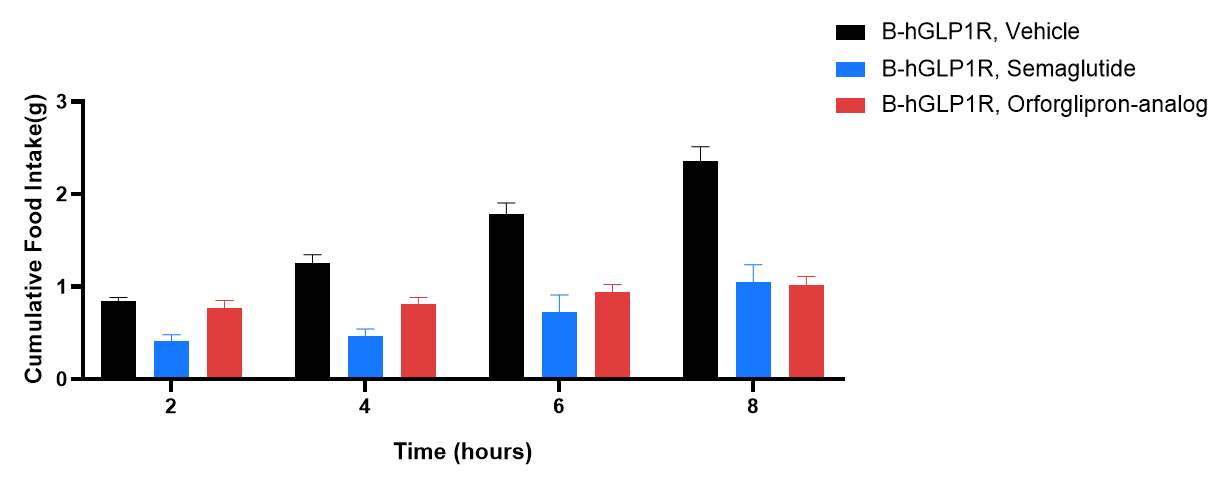
Study of Orforglipron and Semaglutide in B-hGLP1R mice food intake. B-hGLP1R mice (6-7 weeks old) were acclimated for QD PO dosing with vehicle for 4 days, and mice were grouped by body weight and daily food intake. All animals will be placed in clean cages and fasted overnight, mice will be dosed with vehicle or test compound in the morning (before 10 AM). All group's food will be added 15 minutes post-dosing, and food remaining will be recorded at 2-, 4-, 6-, and 8 hours post-dosing. Values are expressed as mean ± SEM. Significance was determined by the 2 way ANOVA. (*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001).

Efficacy study of Semaglutide in HFD induced B-hGLP1R mice (study ID:23P042001). B-hGLP1R mice were fed with a high-fat diet for 12 weeks to induce mice obesity. (A) Body weight change after HFD induction. (B-D) Body weight change after Semaglutide treatment. (E-F) Effect of semaglutide on food intake. 8-10 mice per group. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA. *p<0.05, **p<0.01,***p<0.001 ,****p<0.0001.
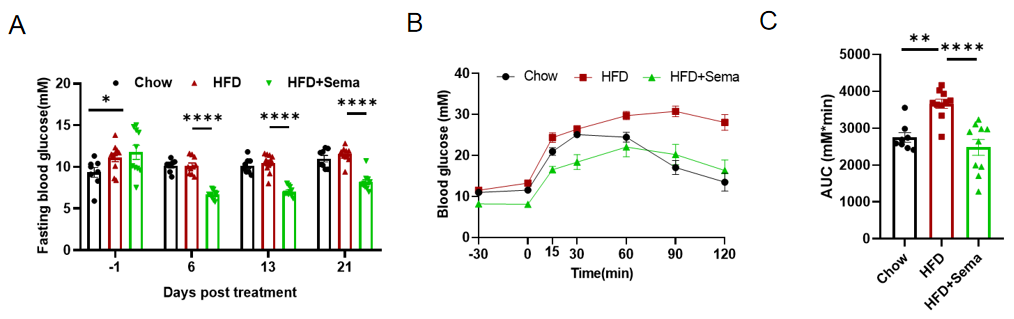
Efficacy study of Semaglutide in HFD induced B-hGLP1R mice. (A) Blood glucose change after Semaglutide treatment. (B) The glucose tolerance test for 2g/kg D-Glucose is administered by intraperitoneal injection (IPGTT). (C) Area under the curve (AUC) for the IPGTT. 8-10 mice per group. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA. *p<0.05, **p<0.01,***p<0.001 ,****p<0.0001.
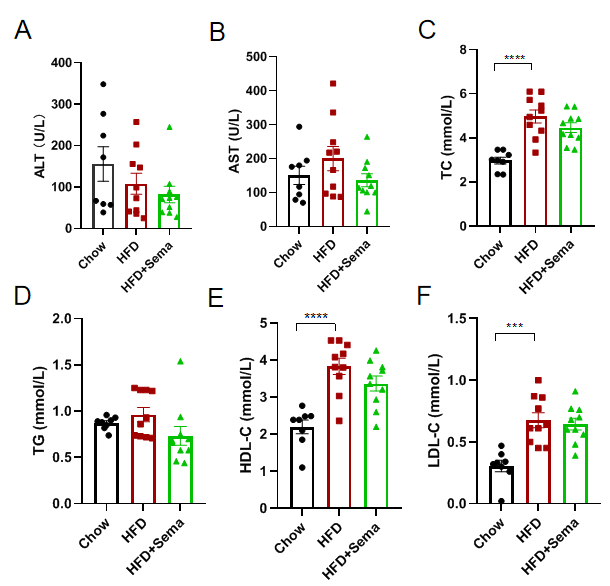
Efficacy study of Semaglutide in HFD induced B-hGLP1R mice. (A-F) Blood biochemical analysis after treatment. 8-10 mice per group. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
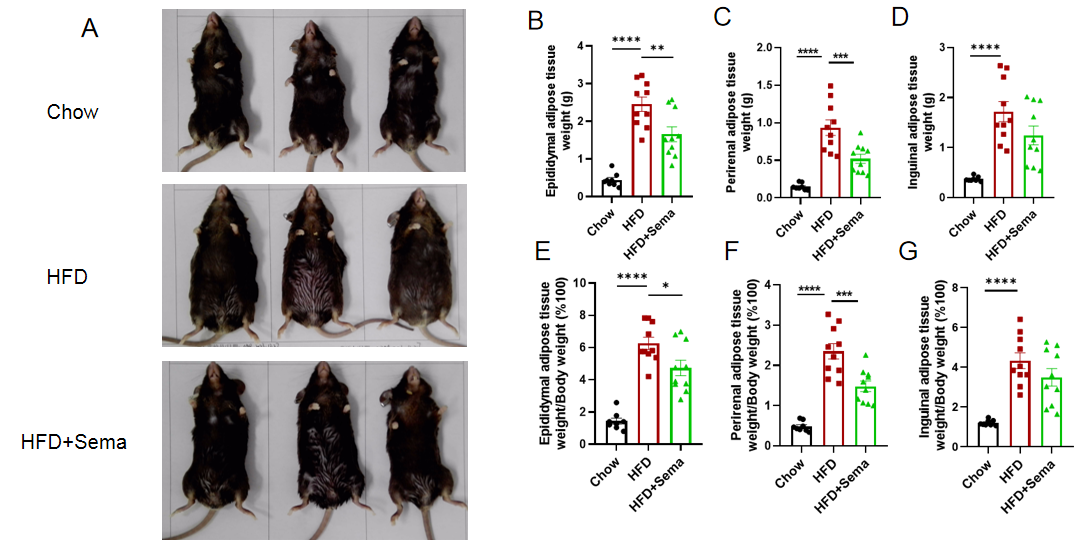
Efficacy study of Semaglutide in HFD induced B-hGLP1R mice. (A) Representative pictures of different groups at termination. (B-D) Adipose tissue weights after treatment. (E-G) Ratios of adipose tissue weight and body weight. 8-10 mice per group. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
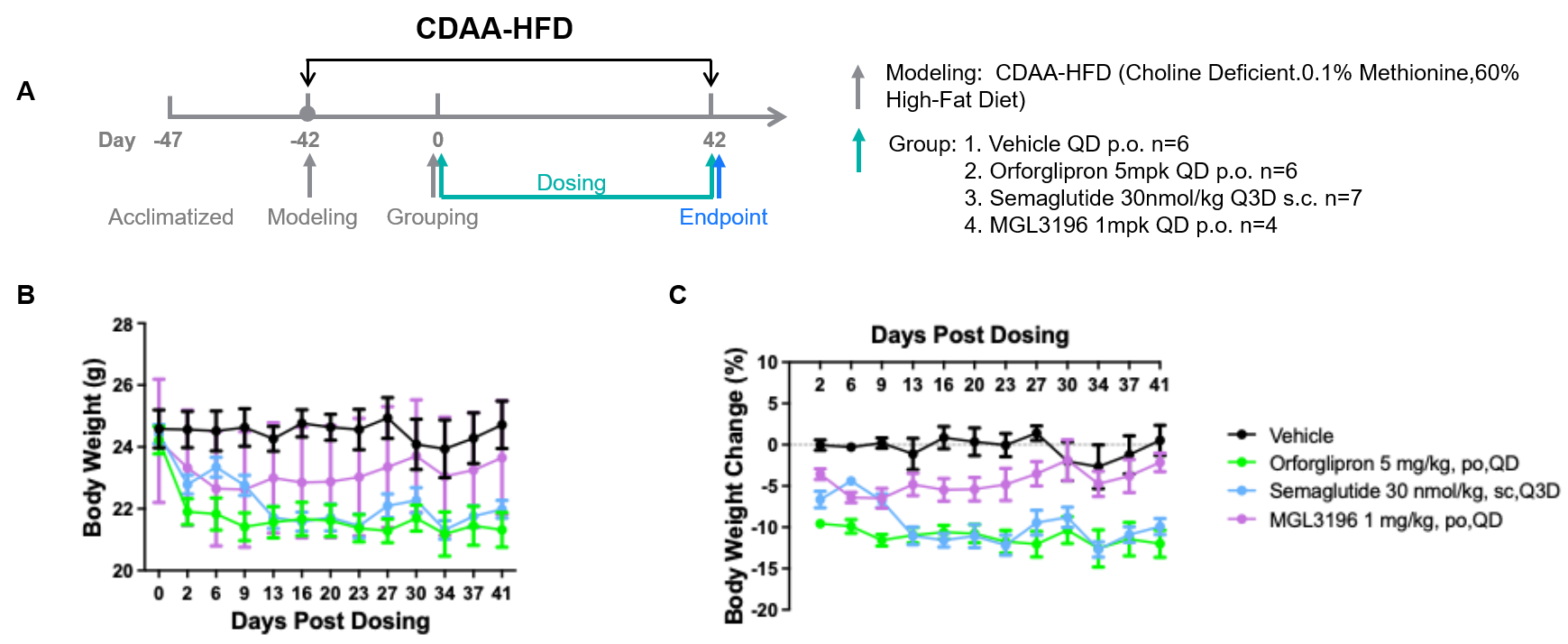
In vivo Efficacy in B-hGLP1R mice CDAA-HFD MASH Model. B-hGLP1R mice were fed with a Choline Deficient.0.1% Methionine, 60% High-Fat Diet to induce CDAA-HFD MASH model. (A) The schematic diagram of experimental processing. (B-C) Body weight change after treatment.
Note: Data was provided by a client.
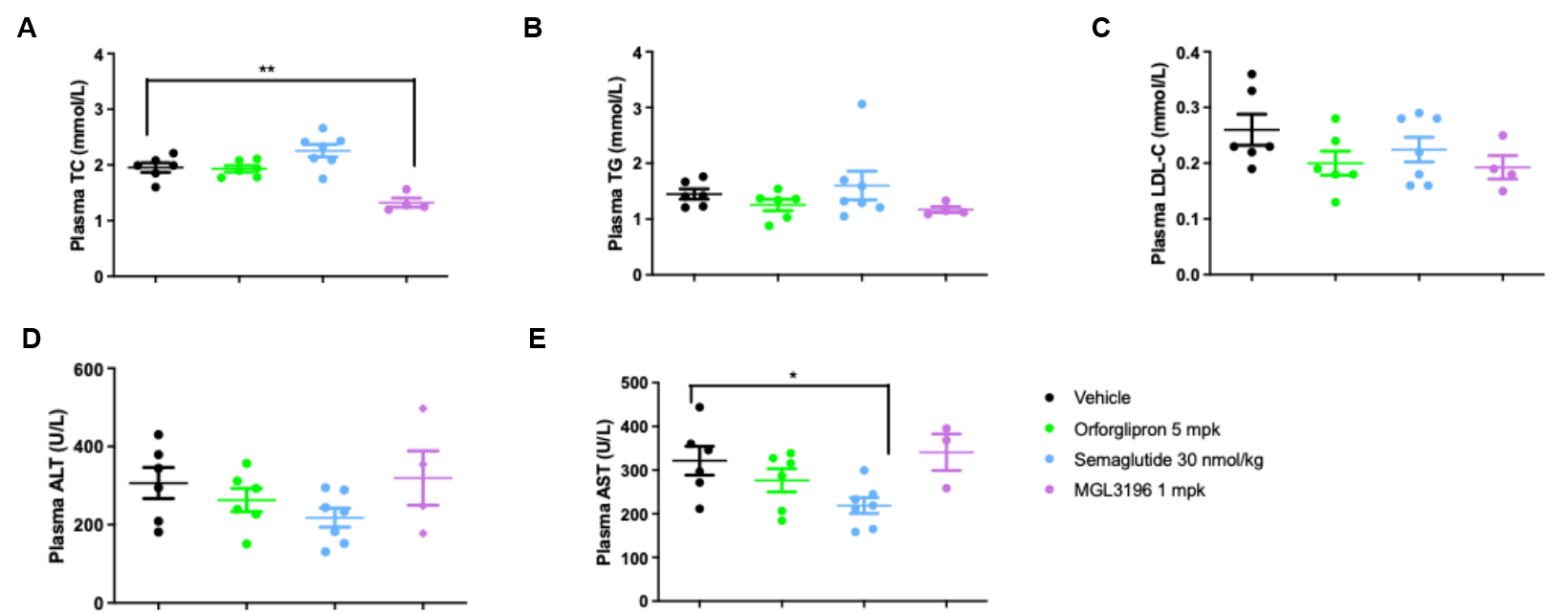
In vivo Efficacy in B-hGLP1R mice CDAA-HFD MASH Model. B-hGLP1R mice were fed with a Choline Deficient.0.1% Methionine, 60% High-Fat Diet to induce CDAA-HFD MASH model. (A-E) Blood biochemical analysis after treatment. Values are expressed as mean ± SEM. Significance was determined by the Ordinary one-way ANOVA. *p<0.05, **p<0.01, ***p<0.001.
Note: Data was provided by a client.
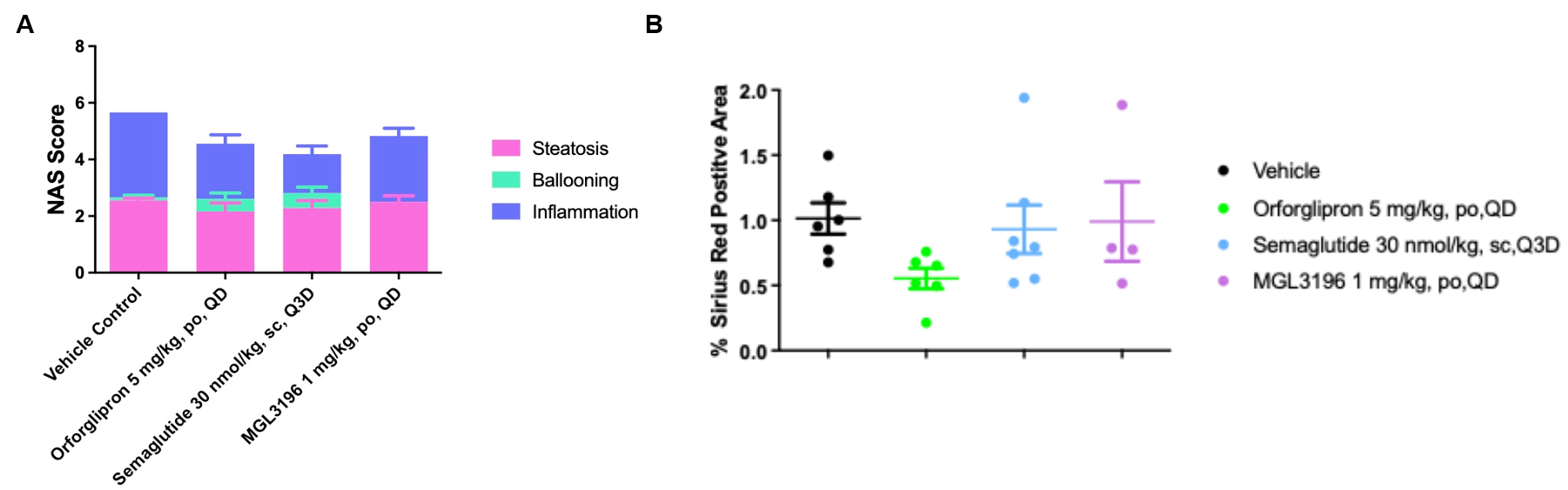
In vivo Efficacy in B-hGLP1R mice CDAA-HFD MASH Model. B-hGLP1R mice were fed with a Choline Deficient.0.1% Methionine, 60% High-Fat Diet to induce CDAA-HFD MASH model. (A) Histological assessment of the NAFLD activity score (NAS), steatosis score, ballooning score, and inflammation score. (B) % Sirius Red Positive Area.
Note: Data was provided by a client.
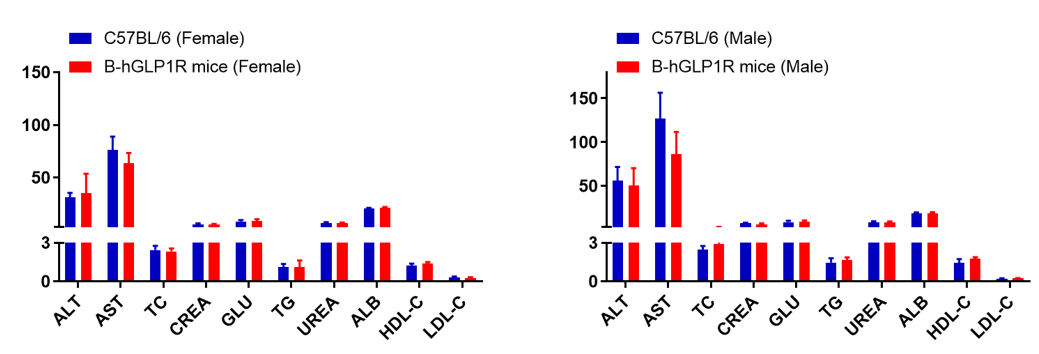
Serum from wild-type and GLP1R humanized mice (n=6, 6–8 weeks old) was analyzed. No significant differences were observed in biochemical indicators between humanized and wild-type mice. Values expressed as mean ± SEM.
Q1: What are GLP1R humanized mice?
A1: GLP1R humanized mice are genetically engineered models in which the murine Glp1r gene is replaced with the human GLP1R gene. These mice express human GLP-1 receptor (GLP1R) in physiologically relevant tissues such as pancreatic islets and gut endocrine cells, while maintaining normal immune and metabolic function.
Q2: Why are GLP1R humanized mice important for diabetes and obesity research?
A2: GLP1R is the therapeutic target of GLP-1 receptor agonists (GLP-1RAs), including Semaglutide and Liraglutide. GLP1R humanized mice provide a reliable preclinical model to evaluate drug efficacy, safety, and pharmacokinetics in vivo, supporting the development of treatments for type 2 diabetes, obesity, and metabolic syndrome.
Q3: Can GLP1R humanized mice be used for antibody validation?
A3: Yes. Because these mice express human GLP1R, they enable in vivo antibody validation and testing of GLP-1 receptor–targeted biologics. Researchers can assess antibody binding, efficacy, and safety in a translationally relevant system.
Q4: How do GLP1R humanized mice differ from wild-type mice?
A4: Unlike wild-type C57BL/6 mice, GLP1R humanized mice express human GLP1R exclusively. Importantly, their immune cell distribution, blood biochemistry, and baseline metabolic parameters remain normal, ensuring that experimental results are attributable to human GLP1R function rather than systemic alterations.
Q5: What disease models can be established using GLP1R humanized mice?
A5: GLP1R humanized mice have been validated in: